In my opinion, Web3 and longevity are the two most promising frontiers. VITADAO sits right at the intersection of the two.
In today’s post, I’ll be talking about VITADAO, which is building a new model for drug development in the Web3 space. But the post is not only on VITADAO - it also covers the problems with the current model of drug development and what the future of science funding could look like.
It’s a long post, but I’ve tried to be comprehensive and explain why an organization like VITADAO exists. I hope that you walk away with a much better understanding of the problems in the drug development process, the incentives of pharma companies & scientists, and how Web3 & VITADAO could be a solution to these problems.1
Welcome to Live Longer World!
Hi everyone! If you are new here, my name is Aastha and I am the creator of Live Longer World.
Through this newsletter, I write on longevity science & health optimization
I am also the host of the Live Longer World Podcast, where I interview scientists, biotech entrepreneurs, and other advocates in longevity science
Previous guests have included Matt Kaeberlein (Rapamycin), Morgan Levine (Epigenetic Clocks), Reason from Repair Biotech, Dave Gobel (Methuselah Foundation) + more!
Find the podcast on YouTube, Apple Podcasts, Spotify, or elsewhere
You will find all my work on my website: livelongerworld.com
My twitter is @aasthajs
My Instagram is @aasthajs
My Patreon is patreon.com/livelongerworld
VITADAO: Web3 x Longevity
Notes on reading this post:
This post has 6 parts. See “Preview of Post” section for topics covered
Each part can be read standalone. For example, if you wish to only learn about VITADAO, read Part 3. But reading through the entire post will give you a deeper understanding of what’s really going on with drug development and the problems VITADAO is trying to solve
Each part (except Parts 5 & 6) has a summary at the end, so should you wish to go back to the gist of each part, you can go back to the summary
Summary of the entire post is included at the end2
If you have any feedback on the post or find any errors that I could correct, please feel free to DM me or email me at livelongerworld@gmail.com
If you are reading this post in your email, I’d suggest reading it on your browser as the email will be truncated due to the length of the post
Preview of Post:
Introduction
Part 1: Current Model of Drug Development
Translation of basic science to clinical science
How does one go from Basic science to Clinical Science?
What are the current sources of funding for clinical research?
Part 2: Problems faced by the current model of drug development
Valley of Death: Basic Science not being translated to Clinical Science
Major Players in the Space & their Incentives
How do pharma companies make money?
Problem #1 Misallocation of Funding
Problem #2 Funding only for “big journals”
Problem #3 Lack of Information Sharing
Part 3: VITADAO, VITADAO FAQ, VITADAO ARCHITECTURE
All your questions on how VITADAO works!
Part 4: How is VITADAO solving the current problems in drug development?
Solution #1: VITADAO has skin in the game in the investment! Further its democratic process serves as a check to misallocation of funding
Solution #2: VITADAO accepts proposals from anyone giving any translational project an opportunity to be funded
Solution #3: VITADAO will strive to make data public
Part 5: Open Questions on VITADAO
Part 6: Bonus - How you can gain from VITADAO + Tidbit on the history & future of asset classes
Let’s begin!
INTRODUCTION
To most people, the concept of crypto, blockchain, and Ethereum seem fuzzy.
To most people, the concept of reversing aging and extending lifespan also seem fuzzy.
What happens when you combine crypto & blockchain with longevity?
Double the fuzziness?
Double the science fiction?
You might think “Sounds cool bro.”
Or you might think “Sounds futuristic & exciting. I want to hear about it!”
If you’re in the 2nd camp, strap on. I’m about to explain the future of longevity and science funding in a manner that will require no prerequisite knowledge of blockchain or longevity.
If you’re in the 1st camp, think again. Before you dismiss this as science fiction, you might want to read the piece to understand why some people are excited about this new model of science funding.
Okay, so what is this new model of science funding? Who is the new guy on the block?
Enter VITADAO.
VITADAO’s mission is “to extend human lifespan by researching, financing, and commercializing longevity therapeutics in an open and democratic manner.”
Their website reads:
“VitaDAO is a new cooperative vehicle for community-governed and decentralized drug development. Our core mission is the acceleration of R&D in the longevity space and the extension of human life and healthspan. To achieve this, VitaDAO utilizes a combination of novel governance (DAOs), digital assets (NFTs) and financial market frameworks (AMMs).”
There’s a lot going on in that paragraph. I’m sure you already have a million questions like:
What is “decentralized drug development”?
What are “longevity therapeutics”?
Can someone explain the last sentence “VITADAO utilizes a...AMMs” in plain English??
Why is their mission to research and finance longevity therapeutics in an open manner? What’s wrong with the current model?
These are great questions. And I’m going to unpack the meaning of all of this. To answer these questions and truly understand VITADAO’s mission, we need some context.
To be specific, we need some context around the current models of drug development, financing of therapeutics, and the problems associated with this model.
PART 1: CURRENT MODEL OF DRUG DEVELOPMENT
Translation of Basic Science to Clinical Science
For a drug to come to market, it requires several steps.
But a simple way to put it is that the basic science embedded in research papers needs to be translated to clinical science.
Basic Science -> Clinical ScienceNow, what does that mean? Let’s use a company building analogy.
We are all familiar with the fact that if you want your idea to be out in the public and add value to potential customers, you need to execute on the idea. A simplified way of saying that is:
Idea -> ExecutionThe execution phase could involve several steps including different iterations, but at this point the idea is not merely in your head. Instead you are testing it with different people and should there be interest in the product you are selling, you can consider commercializing it.
A similar process is happening in scientific labs as it pertains to drug development.
Basic Science -> Clinical ScienceThe basic science is the idea or the research that has been laid out in theory. Is there a way to execute on this science into practical uses such as developing drugs that patients could use to their advantage? Possibly.
And that’s what goes on in clinical science - the basic science is tested in animal and human models to see if there is potential for developing effective cures for patients.
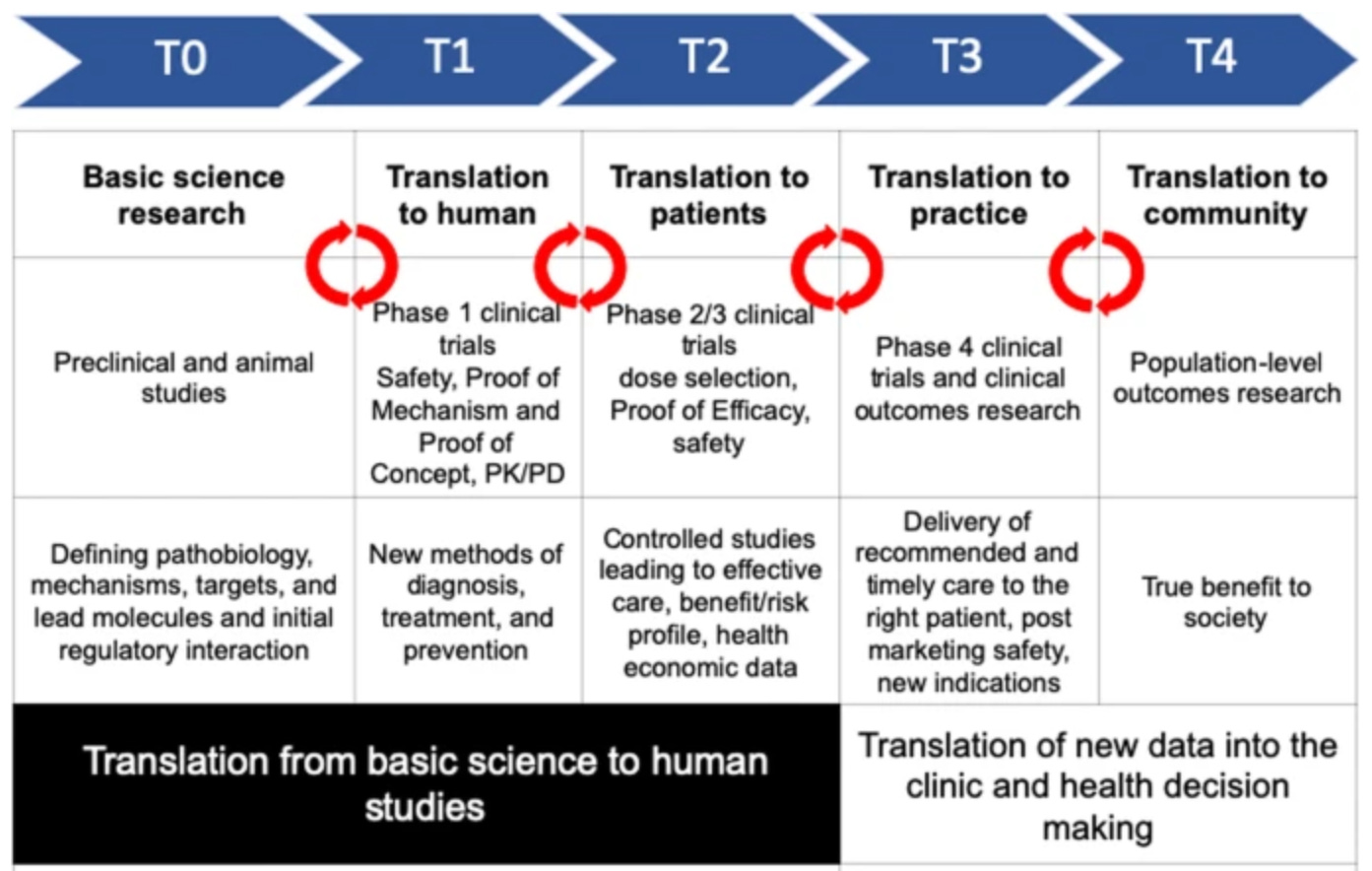
Merely writing basic science -> clinical science is a compact way of saying it. The translation requires several steps. For a fuller picture of the translation, here is the flow chart3:
How does one go from Basic science to Clinical Science?
There are several steps that need to be done in order for basic science to advance to the clinical science phase. These include proof of concept, proof of efficacy, funding, reproducibility of studies etc.
One important requirement for going from basic science to clinical science is funding.
This is similar to how companies and startups sometimes need venture capital and other funding to go from idea to execution.
However, there is an important distinction between funding for (technology) startups and funding for biotech (drug development / translation of basic science to clinical science)4
When it comes to non-biotech company building, it has increasingly become easier and easier to take your idea to the execution phase. With a laptop and some code, you can ship a software product to several potential customers. Or with a cheap microphone and a camera, I can start my podcast. Server costs have gone down, low-code tools are available, and technology has democratized reaching your consumers.
As such, when it comes to company building, the friction in taking your idea to the execution phase has significantly reduced.5
Alas, the same cannot be said for drug development or going from basic science to clinical science.6
Clinical research can require large amounts of funding. Estimates show that the development of a newly approved drug costs about $2.6 billion.7
What are the current sources of funding clinical research?
Most of the money for funding clinical trials comes from biotech Venture Capitalists (VCs) and pharmaceutical companies. Estimates show that pharmaceutical companies spent $83 billion on R&D for drug development in 2019.8 Increasingly, pharmaceutical companies will tap into research projects happening at universities and will fund the research in exchange for Intellectual Property or IP (we will dive into this in Part 2).
SUMMARY OF PART 1: CURRENT MODEL OF DRUG DEVELOPMENT
For drug development to occur, it requires the translation of basic science to clinical science. This means that the research laid out in scientific papers needs to be proven through animal experiments and human clinical trials. In other words, the idea needs to be tested and executed on.
Among other requirements, this translation of research requires large amounts of funding. Currently, most of the funding for clinical trials comes from biotech VCs and big pharma.
PART 2: PROBLEMS WITH THE CURRENT MODEL OF DRUG DEVELOPMENT
Valley of Death: Basic Science not being translated to Clinical Science
As I explained in Part 1, in order for a drug or therapy to reach the market, the basic science must be translated to clinical science.
If there are any errors in translation of basic science to clinical science, the entire drug development process fails. As a result, it’s critical that the translation process doesn’t fail due to errors that are avoidable.
So what happens when promising discoveries fail to be translated into clinical science? They meet the Valley of Death. (Quite a befitting phrase, ha?)
The “Valley of Death” is well-known in science as the place where certain promising discoveries / preclinical findings meet their demise. It’s the obstacles that prevent the basic science from being translated into clinical science.
There are several reasons why discoveries could enter the valley of death - lack of funding, lack of information sharing across scientists, misalignment of incentives of pharma companies, lack of reproducibility of data due to poor experimental designs and flaws in methodology.
As Leo Tolstoy says in Anna Karenina,
“Happy families are all alike; every unhappy family is unhappy in its own way.”
Similarly, the process of translation of basic science to clinical science could be halted for one of several reasons I laid out.
While I won’t touch upon each reason, I’ll discuss some of the key ones that are halting the translation of basic science to clinical science and how VITADAO is solving some of these problems. But before that, you might be asking a question:
Perhaps the basic science that meets the Valley of Death fails for a good reason and is not worth translating to clinical science? After all, not all ideas are good.
You are absolutely right to ask this question. Yes, I’m sure a lot of the basic science that finds its place in the Valley of Death is for legitimate reasons - the idea wasn’t good, the experiment was flawed or not reproducible etc. In cases where the scientific explanation wasn’t worth advancing to the experimental testing phase due to a poor explanation, it’s only fair for that explanation to meet the valley of death.
However, in a lot of situations, the translation fails not because the scientific explanation is not robust enough, but because of external factors such as misallocation of funding, misalignment of incentives and lack of information sharing.
It is these external factors that I’ll be covering.
When the reasons are external in nature, we must ask, is there a way to solve these problems? Why are these problems occurring in the first place?
To understand the causes of the problems around funding and misalignment of incentives, we should take a look at the players in the space and what they are incentivized by.
Major Players in the Space & Their Incentives
As you can see, there are 3 main players involved in the drug development process - researchers, patients, and investors.
Researchers: Scientists, by and large, will try to follow rigorous scientific practices and aim for purity in their research. However, it is possible for scientists to fall into the trap of being incentivized by the attention and profit that comes from publishing in top journals, at the expense of staying true to their research. This desire to publish in top journals is exacerbated by the fact that scientists’ careers are mostly only measured in publications, not medications brought to market.
Top journals sometimes tend to publish studies that fit in a little too neatly, leaving out important pieces of information that do not corroborate with the study findings. In this manner, scientists looking to publish in top journals may sometimes fail to report negative data associated with the study.9
Patients: Patients are consumers who want low-cost and effective therapies and drugs that will cure their illnesses.
Biotech VCs / Pharma Companies: They are one of the biggest funders of clinical trials. Their intention is to bring a drug to market and profit off of it. As such, pharma companies are incentivized by profit and creation of customer value. In fair business practices, creating customer value creates profits. The two are not distinct.
However, there are instances when there can be a disconnect between customer value and profit. Such a situation results in pharma companies chasing profit at the expense of customer value.
What are these situations and why do they occur? We will explore these situations in the next section, but in order to understand why they occur, we must take a brief look at where the returns in biotech investing come from.
Please note that going forward, I’m going to focus on pharma companies as the largest funders of clinical trials, although I presume that biotech VCs would have the same incentives as pharma companies.How do Pharma companies make money & generate returns?
Pharma companies fund clinical trials with the intention of bringing a drug to market that will benefit consumers and make them money. When a promising discovery is made, it is typically patented and the holder of the patent gains access to Intellectual Property (IP) rights.
Intellectual property rights or IP rights give the creator exclusive rights over the creation for a certain period of time. This means that only the holder of the IP has access to commercialize the information produced from the research creation.
So if Pharma Company X decides to invest in Project A, Pharma X will get access to the Intellectual Property filed by Project A, which can later be commercialized to generate money. Commercialization here means bringing a drug therapeutic to the market which people would pay for.
Typically, pharma companies generate most of their profits from selling drugs that are on-patent / have IP rights attached to them. This is because when the drug is on-patent, no one else can sell the drug, so the pharma company can price the drug at a higher price than if there was more competition.
Pharma companies tend to get (a somewhat misguided) bad reputation for selling on-patent drugs at high prices. It’s worth noting a couple of points here:10
Profits from selling high-priced drugs that are on-patent allow pharma companies to keep funding risky clinical trials and drive innovation in healthcare
We should acknowledge that pharma companies take on enormous amounts of risk when funding these trials - some estimates show that the process of getting a new drug from the discovery phase to the market can take more than 13 years and almost 95% of the drugs entering human trials fail.11 Further, as we’ve seen, funding clinical trials is expensive.
As such, profits from selling on-patent drugs allow the pharma company to plough back more money into funding clinical trials
Once these drugs go off-patent, they become “generic drugs” and are sold by several companies for significantly cheaper prices, benefiting millions of consumers
I’m not saying that pharma companies are always good players and there is no price gouging that occurs. Yes, there are cases of price gouging for off-patent drugs.12 But selling on-patent drugs for high prices is not necessarily unethical - it’s sometimes needed for pharma companies to have a sustainable business model
With this backdrop, it will be easy to understand the problems that plague the current drug development process and some of the reasons promising trials meet the valley of death.
Problem #1: Misallocation of Funding - Funding Towards Drugs that are more likely to succeed regulatory approvals, marketing, and generate more profit dollars.
As we’ve already seen, pharma companies are the largest funders of biotech research and are driven by creating customer value and profit dollars. However, when the incentives skew towards profit creation at the expense of customer value, we see a misallocation of funding towards clinical trials aimed at driving large profit dollars and less customer value.
Funding Drugs that are likely to succeed regulatory approvals
Let’s understand this point using an example. Say a pharma company is deciding between funding for development of drug A vs. drug B. Say both drugs confer benefits to patients but drug B has higher benefits. However, for various reasons, drug B might be harder to get approved by the FDA13. Drug A might also have better marketing around it. Big pharma, driven by profit, will likely fund the commercialization of drug A. This could halt further research on drug B even though it could have had a higher impact.
Quoting a research paper:14
Pharma companies are “more interested in focusing on clinical research on candidate drugs that are more likely to succeed the regulatory approvals, and marketing.”
Less Funding for Drugs that are Off-Patent
Given that pharma companies generate profits if they can patent the drug, there is less funding that goes around for discoveries & drugs that are already off-patent but could benefit from further trials being conducted. For example, Rapamycin has shown tremendous promise in improving healthspan & lifespan in mice. However, there isn’t much funding that flows into further trials for Rapamycin and one of the reasons might be that it's already off-patent so it's hard to generate money from it.
Problem #2: Funding only for “big journals”
“The issue with what gets attention is what gets funded, what makes it to the best journals etc. And this is the data people harp on instead of finding the truth.” - Matt Kaeberlein
Another study says:15
“The promotions in basic scientific research have been judged by number of funded grants based largely on the scientific publications in top journals, not on how much they have advanced the clinical science and medicine.”
It turns out science is not immune from human marketing games. Studies that tend to get funded are the ones that get published in top journals. As such, it could be the case that a lot of promising basic science does not get translated to clinical science merely because the science didn’t get published in a top journal!
Not being published in a top journal does not mean the science does not deserve attention.
As an aside, publishing in top journals sounds similar to Ivy league credentialing. Just because one didn’t get into an Ivy league does not mean they aren’t capable of the job. Conversely, just because one went to an Ivy league does not make them capable of doing the job. However, the sad truth is that society still falls for these credentialing traps whereby if you have an Ivy league or top tier school stamp on your resume, you are likely to get more attention for jobs than another candidate who might be equally or more qualified for the job but doesn’t have an Ivy league degree.16
Anyway, a similar process is happening when it comes to publishing in top journals. Studies that get published in top journals receive the most attention and the most funding, while other promising studies not published in top journals meet the valley of death.
Secondary Effect of Problems 1 & 2: Researchers work on projects that are more likely to receive funding as opposed to working on more ambitious projects
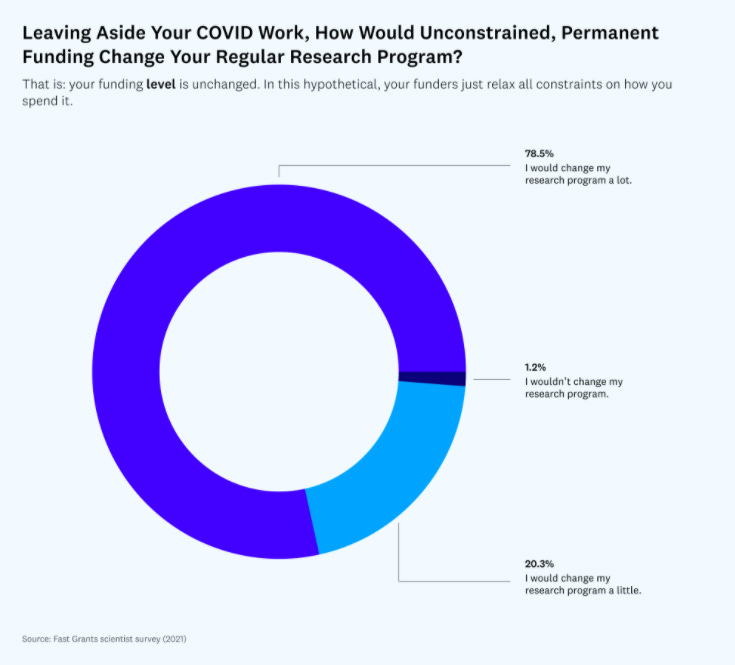
Researchers need funding in order to conduct their studies. Given that pharma companies will tend to publish studies that are published in big journals and those that are easier to succeed regulatory approvals, researchers are sometimes (or oftentimes) forced to work on studies that fit in with this model and will get them funding. As part of their Fast Grants project, Patrick Collision and Tyler Cowen conducted a study17 asking researchers how unconstrained permanent funding would change their regular research program. I was shocked to learn of the results of the survey.
More than 78% responded saying that they would change their research program a lot!
“81% percent of those who responded said their research programs would become more ambitious if they had such flexible funding. 62% said that they would pursue work outside of their standard field (which the NIH explicitly discourages), and 44% said that they would pursue more hypotheses that others see as unlikely (which, as a result of its consensus-oriented ranking mechanisms, the NIH also selects against).”
This survey seems to focus on NIH funding but I don’t see why it would be different if we swap NIH funding with pharma funding.
Consider the implications of these funding mechanisms. They are geared towards funding science projects that are more likely to succeed and prevent scientists from working on ambitious projects. This risk averse nature of funding stifles innovation in biotech and science.
Problem #3: Lack of Information Sharing
Pharma companies are looking to generate profits and compete with one another. As such, when they fund projects, they have little incentive to share data which could prove valuable to other research labs.
Furthermore, pharma companies are particularly disincentivized to share negative research data because they want to downplay the side effects in order to get their drugs approved by regulatory bodies. It is also possible for scientists to be incentivized to leave out messy negative data that doesn’t fit in with a neat story. We’ve noted how top journals sometimes look for simple stories. So scientists looking to publish in these journals may leave out important pieces of information.
Not sharing some of this valuable information often fuels the reproducibility crisis since experiments cannot be replicated if the complete information hasn’t been shared!
SUMMARY OF PART 2: PROBLEMS WITH THE CURRENT MODEL OF DRUG DEVELOPMENT
There are several problems with the current model of drug development and translation of basic science to clinical science. Some of these problems stem from the way the incentive structures are set up for investors in biotech research (pharma companies) and scientists conducting the research. Pharma companies can get biased towards generating maximum profit at the expense of maximum consumer value. One problem is that there is misallocation of funding towards trials that are more likely to generate higher profits and succeed regulatory approvals over trials that potentially lead to maximized patient benefit. Given that pharma companies make money by commercializing a patented discovery, there is less funding towards promising drugs that are off-patent. Two is that science is not immune from human marketing games and most of the funding is only available for studies that are published in top prestigious journals. Promising studies published in “not-so-prestigious” journals tend to not receive funding and meet the valley of death. It is also not always the case that studies published in top journals are rigorous in nature. In fact, top journals will at times tend to promote studies that fit in a little too neatly and leave out important pieces of negative information that do not corroborate with the research findings. A secondary effect of the problems around misallocation of funding is that scientists tend to work on studies that are more likely to receive funding as opposed to more ambitious projects. Three is that trials and studies fail due to lack of information sharing among scientists and pharma companies being disincentivized to share negative research data in order to maximize chances of drugs being approved.
Misallocation of funding, funding biased towards work published in prominent journals, and lack of information sharing are some of the problems that plague the current drug development process and lead to studies meeting the Valley of Death.
In Part 4, I’ll revisit each problem and how VITADAO could be a solution to tackling each of them. Before that, in Part 3, let’s dive into understanding VITADAO, their infrastructure, and what they are all about. But in my mind, it’s more important for you to understand the problems that VITADAO is trying to solve. Once you grasp the problems with the current model of drug development, you will have a higher appreciation for truly understanding VITADAO and why they are deploying their unique infrastructure as a solution. So let’s jump in!PART 3: VITADAO, VITADAO FAQ AND VITADAO ARCHITECTURE
Very high-level, what is VITADAO?
VITADO is a DAO (decentralized autonomous organization) committed to funding early stage longevity research in a democratic manner.
What is a DAO / decentralized autonomous organization?
The simplest way to explain DAO comes from the Ethereum.org website, which says DAOs are:
Member-owned communities without centralized leadership.
A safe way to collaborate with internet strangers.
A safe place to commit funds to a specific cause.
“Think of them like an internet-native business that's collectively owned and managed by its members. They have built-in treasuries that no one has the authority to access without the approval of the group. Decisions are governed by proposals and voting to ensure everyone in the organisation has a voice.
There's no CEO who can authorise spending based on their own whims and no chance of a dodgy CFO manipulating the books. Everything is out in the open and the rules around spending are baked into the DAO via its code.”
In one sentence, VITADAO is member-owned and funds longevity research in a democratic manner.
Isn’t that cool??
Isn’t it amazing to be part of an organization that is owned by its members? And no crazy boss can come along and change the rules of how the organization functions? You get to be part of each decision by voting on them and everything is decided in a democratic manner.
That was a high-level overview of VITADAO. Let’s dive into some of the intricacies (explained simply!) using an FAQ format. VITADAO FAQ:
What is VITADAO’s mission again?
VITADAO’s mission is “to extend human lifespan by researching, financing, and commercializing longevity therapeutics in an open and democratic manner.”
How can I become a member of VITADAO?
You will become a member of VITADAO if you hold VITA tokens. These are governance tokens that give you voting power.
You can own VITA tokens by purchasing them or by contributing work to the community.
Remember, it's an organization owned and run by its members. Members ensure the smooth functioning of VITADAO by contributing useful work such as code, content, proposals etc.
In return for their contribution of work, they are rewarded with VITA tokens.
How can I purchase VITA tokens?
Uniswap or balancer. Link here.
How can I contribute work to VITADAO?
VITADAO has different working groups such as Governance, Tokenomics, Longevity, Awareness, Legal and Operations.
Depending on your interests, you can join one of the working groups and see how you can begin to contribute work. In fact, VITADAO is open to all kinds of contributions, so if you have a new idea for a working group, you can propose that and create one too!
What do I gain from being a member of VITADAO?
You will be supporting the funding of longevity research in a democratic, decentralized manner! But also, you get to invest in early-stage biotech with the potential to make significant returns on your investment! (See Part 6 bonus section for more on this)
Why is VITADAO funding longevity research?
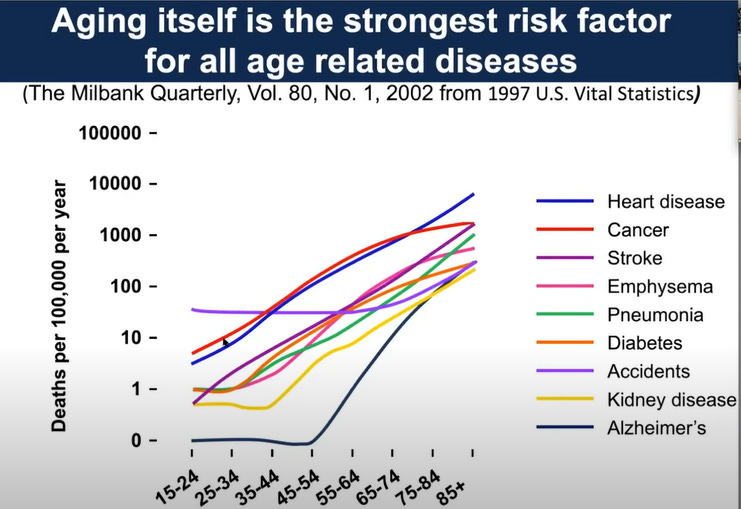
This is another way of asking: why is longevity research important and what is longevity research? Longevity research is directed towards developing therapeutics that can slow or reverse aging. Aging is the biggest reason for the onset of diseases such as cancer, cardiovascular risk, atherosclerosis etc. As such, if we can target the biology of aging itself, we can prevent the onset of diseases and extend human lifespan & healthspan (the number of healthy / disease free years a person has).
And this is not science fiction. The field of longevity science is already showing results and there are brilliant scientists working on targeting the biology of aging to prevent and reverse the damage from diseases.
How are decisions to fund projects made? How is it “open and democratic?”
As a VITADAO member, you own tokens which give you access to vote on longevity research proposals.
Say another member John comes along and says, “Hey, this Project A being done by Lab X looks promising in improving healthspan. Can we consider funding it to advance it to clinical trials?”
In this manner, John introduces the proposal to all the VITADAO members. As a member, you can independently evaluate whether you think the project is indeed promising and worth funding. The community will decide to fund the research project if it passes a majority vote of its members.
As such, the decision to fund a project is decided democratically. There is no “management team” deciding which project to fund and how to allocate money. There is however a working group, the longevity working group, that curates research applications that are voted upon by each member. There are no “senior managers” bringing in projects and proposals. Anyone (member or non-member) can pitch a proposal to the VITADAO members and the community makes the funding decision by voting on the proposal. Every member gets a chance to be part of the decision making process. The decisions are not being made behind closed fancy offices by a few men in black suits.
Great, but what is the path to making money and returns on my VITA tokens?
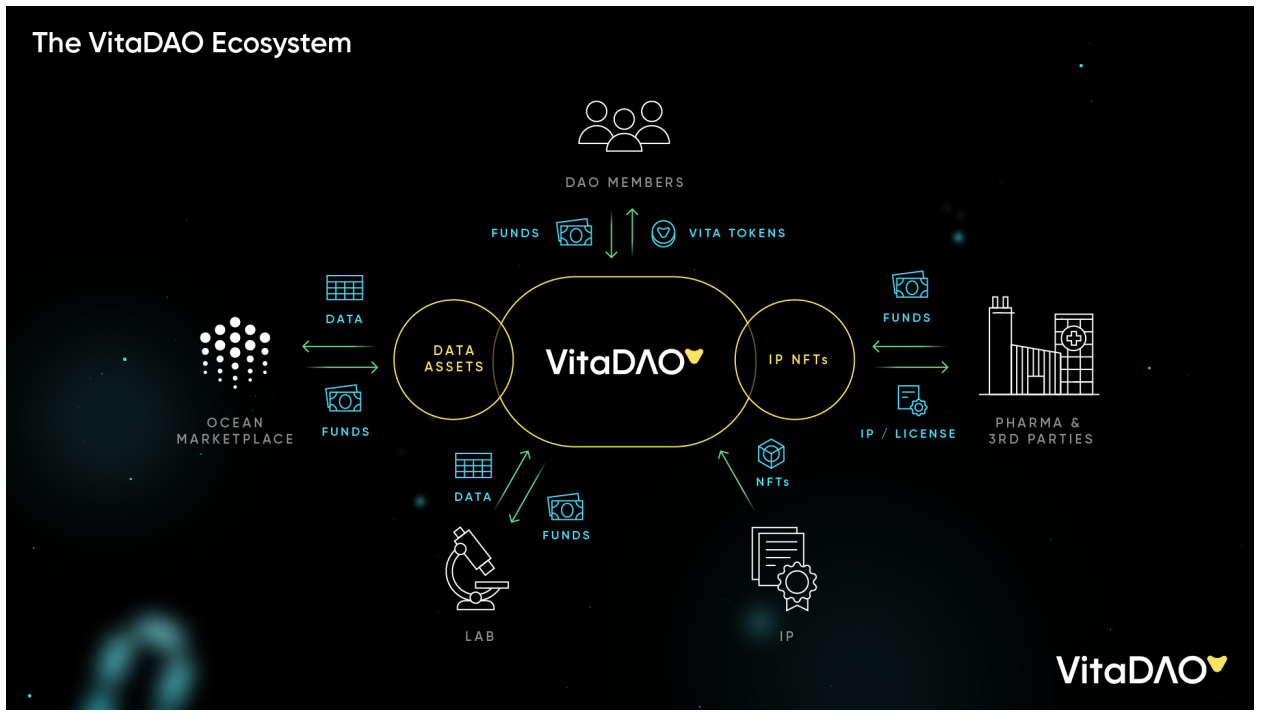
As a VITA token holder, you can make money if your token goes up in value. There are high chances that the value of the token will go up if the DAO treasury grows in value. Essentially, you should care about how VITADAO will generate money and value - because only if it does, it will be a sustainable funding organization. Let’s grasp this by diving into the VITADAO architecture and understanding their ecosystem schematic.
In exchange for funding the research lab to work on a new longevity therapeutic, VITADAO gets access to Intellectual property (IP). We’ve already seen how IP is an asset and can be used to commercialize research and generate profits.
There are notable differences in how VITADAO plans on holding these IP rights compared to the traditional model. These differences could play a critical role in shaping the future of biotech funding. Let’s try to understand it.
IP NFTs:
This is where things start to get cool and maybe confusing for some people. VITADAO will hold IP rights in the form of NFTs - think of it as a digital version of the IP. Molecule is a platform that connects biopharma research to funding and they work on developing an NFT (digital) version of the IP.) Using Molecule’s technology, this IP NFT that originally belonged to the lab can be transferred to VITADAO in exchange for funds. You’re probably wondering why VITADAO will hold the IP in the form of an IP NFT as opposed to a traditional IP. IP NFTs are cool and useful for a few main reasons, two of which I’ve explained below:
Greater liquidity and value add to IP: IP NFTs can be fractionalized to enable more people to participate through investing small sums of money. This would add more liquidity to biotech funding.
As Tony Kulesa of Petri Bio pointed out, “Fractionalizing ownership of IP lowers the bar so that anyone can buy a stake and then contribute to raise the value of the IP, eg sharing new research about how it might be applied, improving upon it (eg insilico screening, med chem, etc), even business development / dealmaking.”
Fractionalizing IP NFTs would essentially mean that there are several owners of the IP, all of whom are invested in raising the value of the IP. Each owner of the IP can contribute in their own way to raise the value of the IP - this could involve contribution through striking deals with relevant partners, helping with the research etc. It’s like there is an army of IP owners batting for the success of the IP.
Greater portability and faster transfer of IP ownership: If you want to sell your IP NFT to someone, you will not only have greater liquidity due to the fractionalization of the IP, but the transfer of ownership will also be quicker using blockchain technology. You won’t have to go through several legal hurdles.
Monetization of IP:
VITADAO can license or sell these IP NFTs to pharma companies or other 3rd parties to generate income. As a plus, IP rights generate access to data produced by research labs as well, which can also be monetized in the form of NFTs.
In summary, through funding research labs, VITADAO gets access to data and IP. The IP will be held in the form of an IP NFT which will allow for greater liquidity and value add through fractionalizing ownership of IP. As an IP-NFT both of these can be sold to pharma companies and other 3rd parties to generate income and enable sustainable science funding.
What happens when there is successful commercialization of IP? Where does the money go?
Members of VITADAO will decide on how to distribute income generated from successful IP monetization. VITADAO whitepaper reads:
“Individual IP and NFTs may be spun out into separate fractionalized markets to enable more granular participation by the public.”
So again, members vote on what should be done with the income. As I touched upon above, one idea could be fractionalizing the IP further to enable more people to participate through investing small sums of money. That said, the DAO does not plan to ever pay dividends to its token holders.
Is there enough liquidity in the VITA tokens trading?
Less liquidity than exists in the public stock markets! You can take a look at liquidity here. But the point of buying VITA tokens is not speculation or day-trading, rather it is to support promising longevity projects and participate in the downstream value creation.
In the near future, if you are willing to “stake” your VITA tokens for a period of time until a project milestone is reached, you will get rewarded for showing your long-term support and foregoing liquidity for a period of time. Staking signals long-term support and confidence in the project, thereby reducing volatility.
How is VITADAO decentralized?
There is no boss or central authority! No one person controls VITADAO. You are a member if you own tokens and you have voting rights in accordance with the amount of tokens you hold. And as we’ve seen above, every member can participate in the open and democratic decision making process.
Sure, but what if one member purchases the majority of the tokens, giving them more voting power?
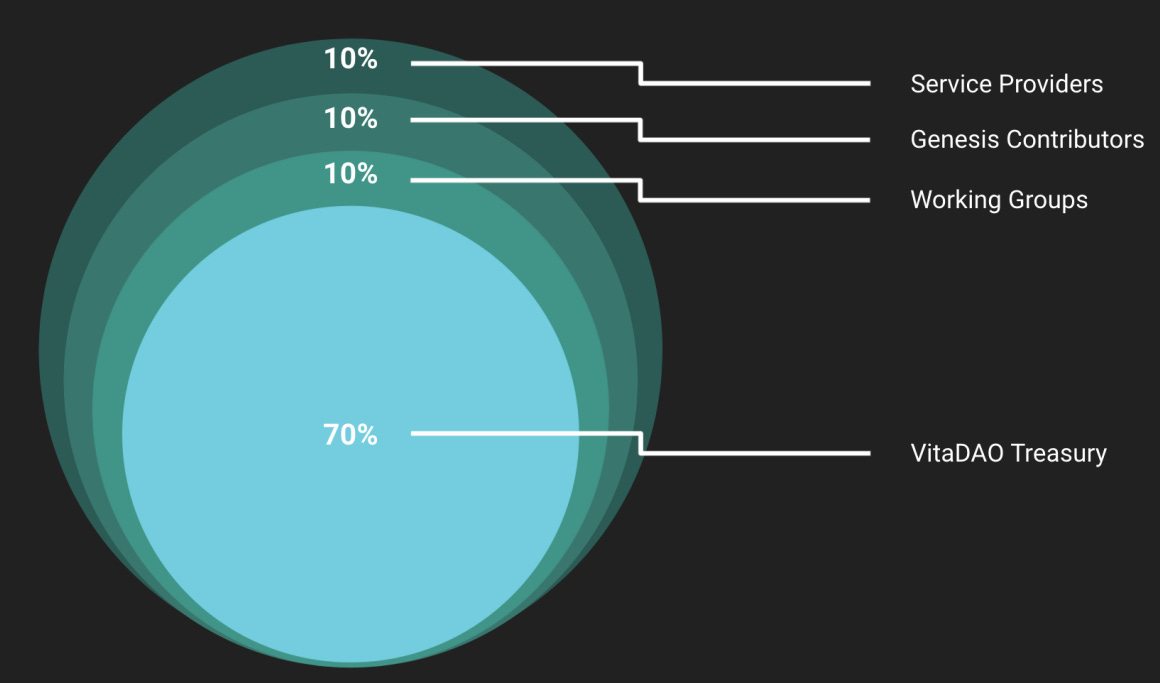
In the first round of token auction (when VITA tokens were first brought to market), only 30% was minted, whereas the remaining 70% remains in the treasury for future projects and is not available to be purchased now. So, at least in this first round, it’s impossible for anyone to hold more than 50% of the tokens since that number is not even in supply!
What else should I know about VITADAO?
It’s an open and friendly mission-oriented community! You can join their Discord without having to purchase VITA tokens. And you can check out their website vitadao.com
Now read their mission statement again - does everything line up and make sense?
“VitaDAO is a new cooperative vehicle for community-governed and decentralized drug development. Our core mission is the acceleration of R&D in the longevity space and the extension of human life and healthspan. To achieve this, VitaDAO utilizes a combination of novel governance (DAOs), digital assets (NFTs) and financial market frameworks (AMMs).
We covered how it’s community owned. We covered how its mission is to fund longevity research. We covered what a DAO is. We covered how its governance is novel through being open, democratic and decentralized. We covered how VITADAO can make money using data IP NFTs to enable sustainable science funding.
We covered a lot. We covered the future.
SUMMARY OF PART 3: VITADAO, VITADAO FAQ & ARCHITECTURE
VITADAO is a member-owned community whose mission is to invest in longevity research. Anyone can be a member of VITADAO by purchasing VITA tokens or by contributing work to the community. It operates with a unique governance structure that is open, democratic and decentralized. Funding decisions for longevity research projects are made by the members through a voting system. If VITADAO decides to fund a research project, it gains access to the data and intellectual property (IP) in the project. VITADAO will hold the IP in the form of IP NFTs. This will allow for fractionalized ownership of IP, facilitating greater liquidity and value-add to the IP. IP NFTs can be commercialized and sold to 3rd parties in order to generate income for VITADAO.
Turns out I’m not yet done. I started my writing by talking about the current model of drug development and the problems associated with it. And I promised you that I’ll show you how VITADAO is solving these problems and why this new guy on the block has a reason to exist in the first place. Strap on - let’s journey through Part 4!PART 4: HOW IS VITADAO SOLVING THE PROBLEMS WITH THE CURRENT MODEL OF DRUG DEVELOPMENT?
Now that you understand the current model of drug development, the problems it faces and the functioning of VITADAO, we can look into why VITADAO might be a solution to these problems. I’ll tackle each of the problems I laid out above.
Problem #1: Misallocation of Funding - Funding Towards Drugs that are more likely to succeed regulatory approvals, marketing, and more profit dollars
Solution #1: VITADAO has skin in the game! Plus, its democratic voting process should serve as a protection against incentive to fund projects that are biased by marketing games
If you recall, I spoke about how misallocation of funding arises when there is a disconnect between customer value creation and profits generated by pharma companies.
With the VITADAO funding approach, the patients who need the therapeutics can be funders of the therapeutics! This is true skin in the game and overlap between customer value as well profit generation.
In the case where the funders of research are not the ultimate beneficiaries, I’m not sure if VITADAO offers a clear solution to this problem. Misallocation of funding by pharma companies is because of improper incentives and decisions being made by a few big players. At the end of the day, VITADAO also wants to generate profits, so there is a risk that the members fall into the same trap as big pharma companies. Further, the applications for funding will be curated by a longevity working group within VITADAO and could be subject to the biases of this group.
Also, what will incentivize VITADAO to fund promising off-patent drugs? They too make money through commercialization of IP rights.
However, the fact that projects are decided by a democratic process sets up the stage in a manner where no one person driven by profit can drive the decision. Given that the community is mission + profit oriented, and includes members who themselves would benefit from therapeutics, I would hope that most of them lean towards making decisions that will be value add for patients.
Problem #2: Funding only for “big journals”
Solution #2: VITADAO accepts proposals from anyone giving any translational project an opportunity to be funded! As such it is diversifying the funding landscape for translational research
This is the bread and butter of VITADAO. Okay, that’s an exaggeration. But VITADAO accepts research project proposals from anyone and the project is voted on by each member. Their filter for accepting proposals is not simply whether the research has been funded in prominent journals. In fact, this is one of the points they harp on in their white paper - that they want to fund good science that tends to get neglected just because it didn’t get published in prominent journals. This is especially relevant in ageing research, which until recently has been underrepresented in major journals. Because as we’ve seen, science is not immune to marketing games and getting published in prominent journals doesn’t necessarily mean the science is robust.
On the flip side, not getting published in prominent journals is not necessarily an indication of less robust science that doesn’t deserve to be funded.
By accepting proposals from anyone, VITADAO eliminates this “fund research published in major journals” bias that is easy to fall into.
Problem #3: Lack of Information Sharing
Solution #3: VITADAO will strive to make data public
VITADAO plans on making data from research projects available to the holders of the IP-NFT. Think of it like the IP-NFT being a key to a “dropbox” type repository with research data rather than simply a signed piece of paper.
This way, data will be available to all who have skin in the project.
By sharing data, VITADAO will allow researchers to build information upon each other. Each building block can add up to an empire! Data sharing can also open up doors to take science research in new directions that perhaps have not even been envisioned before.
The following is a quote from Chris Dixon in a recent podcast with Naval and Tim Ferriss on “The Wonders of Web3”:
“They’ll (Web3 games) be completely composable in that any piece from any other app can plug into any other app permissionlessly. And so you’re building an edifice. It’s almost like building a civilization or a city of interconnected apps instead of these silos in which the data’s not portable, the code isn’t portable, users aren’t portable.”
Chris Dixon is talking about Web 3 games, but something similar can be said about biotech research that is funded by VITADAO and where data is shared openly to IP-NFT holders. If the data can be plugged into different research models, researchers can verify studies, build upon each other’s work and create an edifice!
SUMMARY OF PART 4: HOW IS VITADAO SOLVING THE PROBLEMS WITH THE CURRENT MODEL OF DRUG DEVELOPMENT?
VITADAO is addressing some of the problems with the current model of drug development. As a funding vehicle, they are adding more dollars to investing in longevity research. VITADAO is addressing the problem of misallocation of funding by having more skin in the game and through a democratic process which can serve as a check. The funders of the research can be the ultimate beneficiaries of the research, which is true skin in the game! VITADAO accepts research proposals from anyone and does not care whether the research was published by a big journal or not. Finally, VITADAO will enable data sharing of those data assets that do not compromise with obtaining IP rights. In this manner, VITADAO will make valuable information available to anyone who can benefit from it, make the process more transparent and help in the advancement of science.
PART 5: OPEN QUESTIONS ON VITADAO
Research proposals will be voted on by the members of VITADAO. Do they have the knowledge to know whether the research is worth funding and is promising?
I don’t think the members have to be “experts” when deciding which projects to fund. Experts are fallible too. But you need to be able to judge and think critically about the different research options to fund.
As far as I understand, VITADAO has a scientific community / longevity working group that helps in vetting projects and could assist the members in evaluating projects. But then there is a risk of bias such that the proposals highly ranked by the scientific community might be the ones that tend to get voted on.
One of the problems VITADAO highlights in its white paper is that pharma companies have a tendency to not share negative data. Why can’t VITADAO projects fall into the same trap? What controls are in place?
Perhaps a contract that the IP-NFT comes with access to data?
Is there going to be enough money through VITA tokens to be able to fund multiple projects?
VITADAO aims to commercialize and profit from its initial projects which would generate more money to fund future projects. However, this plan hinges on the successful monetization of the initial projects.
Who is to say that VITA token holders don’t fall in the same trap as pharma companies - funding research that will generate the most profit as opposed to most value to patients?
I spoke about this from a skin in the game perspective. Yes, if the funders are the beneficiaries, this risk is eliminated. However, this risk holds in other cases.
Big pharma is so big that they can afford to lose money on several biotech projects and still be able to fund tons of other ones. It’s like a big VC portfolio. Can VITADAO afford to take such a level of risk?
Here’s my take on addressing this risk: By being member-owned, VITADAO can afford to take more risk since the risk is distributed across members, each of whom takes a small amount of risk in return for the opportunity to generate enormous returns. This is an example of the convexity and antifragile principle popularized by Nassim Taleb. You make high-convexity small bets, each of which could lose you a little bit of money, but if one of them succeeds, you get a huge payoff. However, I’d still like to see how this pans out.
A FEW LAST WORDS ON VITADAO
VITADAO is a longevity focused investment community with a very unique structure:
Anyone can be an investor. Goodbye high-net worth need.
Anyone can showcase their work. Goodbye Ivy-league credentialing.
Anyone can be an equity owner. Goodbye traditional VC funding.
Anyone can introduce a proposal. Goodbye deal scouts.
Anyone can participate in decision making. Goodbye bosses.
Hello future!
PART 6: BONUS SECTION - TIDBIT ON HISTORY & FUTURE OF ASSET CLASSES + HOW YOU CAN GAIN FROM INVESTING IN EARLY STAGE BIOTECH
The history of investing in asset classes outside of public equities is inundated by big companies controlling large swaths of capital and the requirement of having a high net-worth to invest in these asset classes. For example, investing in startups, art, real estate used to be restricted to those who had large amounts of capital. If you didn't have large amounts of money, you simply couldn’t invest.
In the last few years, with the rise of funding marketplaces, this began to change. By allowing for fractionalization of shares and allocating smaller pieces of asset classes to everyone, funding marketplaces have enabled the general public to invest as little as they want in some of these asset classes.
AngelList runs a platform where accredited investors can more easily invest in startup deals they may not have had access to earlier. Marketplaces like Yieldstreet allow everyone to invest small amounts in real estate. Republic allows people to invest as little as $10 in startups and small businesses. Masterworks lets you take small equity ownership in pieces of art. There are lots more examples.
What’s the future?
While these funding marketplaces have vastly improved the landscape for allowing general consumers to invest in asset classes, you still have to depend on these marketplaces to feature the investment deals. For example, if for some reason, Yieldstreet does not decide to list a particular piece of property, you as an investor won’t have an opportunity to even evaluate the deal.
Web3 is changing that. Through vehicles like DAO’s, there is no marketplace or middleman between the investor and the asset looking for investment. Anyone can pitch their idea / venture to the members of the DAO, who decide whether to fund it or not.
I won’t do justice to VITADAO if I don’t reiterate my point on fractionalized ownership of IP-NFTs. VITADAO is allowing you to invest small sums of money in early stage pharma research. There is currently no other vehicle that will let you do that. Investing in early stage pharma has been restricted to biotech VC’s or pharma companies. The best you could do as an outside investor was to invest in public biotech stocks. Now, through VITADAO you can make a bet early on. And if you’ve invested in a promising asset before, you know that the best returns come from being an early investor.
One last point, you get to be a part of the change you wish to see.
“Imagine research on a new breakthrough insulin treatment funded by diabetics who believe in it and stand to benefit the most from it” - VITADAO whitepaper
The future is what the past was supposed to be. For the people, by the people.SUMMARY OF POST:
VITADAO - A NEW MODEL FOR DRUG DEVELOPMENT & BIOTECH FUNDING
VITADAO funds longevity research in an open and democratic manner. Longevity research is directed towards developing therapeutics that can slow or reverse aging. Aging is the biggest reason for the onset of diseases such as cancer, cardiovascular risk, atherosclerosis etc. As such, if we can target the biology of aging itself, we can prevent the onset of diseases and extend human lifespan & healthspan (the number of healthy / disease free years a person has).
VITADAO is a decentralized autonomous organization and anyone can be a member of VITADAO by purchasing VITA tokens or by contributing work to the community. It’s a community that is owned by its members and no one person controls the functioning and decision making.
In exchange for funding longevity research, VITADAO holds Intellectual Property in the research project. The IP will be held in the form of NFTs and decisions to fund the research are made in a democratic manner. This will allow for fractionalized ownership of IP, facilitating greater liquidity and value-add to the IP. IP NFTs can be commercialized and sold to 3rd parties in order to generate income for VITADAO.
There are a couple of reasons for a structure such as VITADAO to exist. To understand the reasons, we must understand the problems with the current model of drug development and the solutions that VITADAO offers.
Drug development requires the translation of basic science to clinical science - which is to say that the science laid out in research papers needs to be proven out through animal experiments and human clinical trials. This translation process requires several processes to align - funding, reproducibility of data, information sharing.
If any one of these processes fail, the development of the drug can fail and the basic science research meets the Valley of Death, i.e. it faces its demise and is not translated into clinical science.
If we care to develop effective therapeutics for patients, it is critical to ensure that the science does not meet the Valley of Death due to errors that were avoidable. What are these avoidable errors and why do they occur?
Some of these errors occur due to misallocation of funding towards drugs that have better marketing behind them, bias towards funding studies only published in “top journals”, and lack of information sharing among scientists.
Why do these errors occur? To understand these, we have to peel into the incentives of the parties involved in the drug development process. There are 3 major parties involved in the process of developing drugs - scientists, investors, and patients.
Patients want low-cost and effective drugs. Scientists want funding for their research, but sometimes they could also be incentivized by profits from commercialization of research. And the biggest investors of biotech research are pharma companies and biotech VCs. Investors are incentivized by the creation of customer value and profits. However, there are times when the incentives can be skewed towards profits at the expense of customer value. In cases such as these, pharma companies tend to allocate funds to projects that have better marketing around them and are more likely to succeed regulatory approvals, and generate profits. There is also more funding towards studies published in top journals, which means that promising studies not published in prestigious journals could be neglected. Lastly, pharma companies are disincentivized to share negative data associated with the studies in order to increase the probability of the study passing regulatory approvals. Without complete sharing of information, it becomes difficult to reproduce studies.
All these errors result in promising studies meeting the Valley of Death, thereby halting the potential of effective drugs to be developed.
So how is VITADAO addressing these problems?
VITADAO is addressing the problem of misallocation of funding by having more skin in the game and through a democratic process which can serve as a check. The funders of the research can be the ultimate beneficiaries of the research, which is true skin in the game! VITADAO accepts research proposals from anyone and does not care whether the research was published by a big journal or not. Finally, VITADAO will enable data sharing of those data assets that do not compromise with obtaining IP rights. In this manner, VITADAO will make valuable information available to anyone who can benefit from it, make the process more transparent and help in the advancement of science.
VITADAO is building the future of science funding! I can’t wait to see how it pans out.
If you’ve gotten this far and read the entire post, thank you and hope you found it valuable! If you have any feedback, you can DM me or email me at livelongerworld@gmail.com. Or feel free to drop it in the comments!
I will be making a video version of this post, so if you want to be notified of it, you can subscribe to my YouTube, which is where I also release video versions of my Live Longer World Podcast.
If you liked the post and learned from it, please share the post and spread the word!
Happy holidays,
Disclaimer: This is my independent analysis of VITADAO. I do not (yet) have any affiliations with them. However, Niklas Rindtorff, who is a core member of the VITADAO community has reviewed the post to check for factual errors as it pertains to VITADAO. He also graciously provided me feedback and valuable input on the entire post.
David Deutsch’s book Beginning of Infinity served as an inspiration for the structure of the post. Off topic, but highly recommend the book - gave me a new perspective on the world!
Source of flow chart: https://transmedcomms.biomedcentral.com/articles/10.1186/s41231-019-0050-7
There are in fact several distinctions between tech and biotech. More reading: https://www.celinehh.com/tech-vs-biotech
With the exception of deep tech and companies that are heavy on physical infrastructure
In the future, I envision that this process will have a lot less friction for clinical trials as well. We’ll be able to experiment easily, not only increasing the volume of experiments we run, but also the time needed to run experiments. In fact, I believe, Niklas from VITADAO is working on an idea like this!
https://dukespace.lib.duke.edu/dspace/bitstream/handle/10161/12742/DiMasi-Grabowski-Hansen-RnD-JHE-2016.pdf;jsessionid=F8E06B62BE6DDD719550D11BA7B587A7?sequence=1
https://www.cbo.gov/publication/57126
Please note that I’m not saying all studies that get published in top journals don’t report negative data or that all scientists can fall into this trap. Of course there are credible studies published in top journals too. But not all studies are true to the data.
For further reading on this, I recommend the book The Great American Drug Deal
https://transmedcomms.biomedcentral.com/articles/10.1186/s41231-019-0050-7
AbbVie’s drug Humira is a good example of off-patent price gouging: https://www.pharmaceuticalprocessingworld.com/house-report-accuses-abbvie-of-price-gouging/
The U.S. Food & Drug Administration is a regulatory body which is responsible for regulating and approving drugs before they can be sold to patients
https://transmedcomms.biomedcentral.com/articles/10.1186/s41231-019-0050-7
https://transmedcomms.biomedcentral.com/articles/10.1186/s41231-019-0050-7
Btw, I’m not trying to hate on Ivy-Leagues. I went to an Ivy-League myself!
https://future.a16z.com/what-we-learned-doing-fast-grants/